 Therefore, the halogen reactivity decreases down the group due to the fact the atomic radius increases and shielding increases down the group as the molecules get bigger as they contain more electron shells, so the attraction between the incoming electron theyre trying to GAIN and the positive nucleus in weaker, so they are unable to attract the electron as easily as higher up halogens which attract the incoming electron with a lot stronger force. 
Reactivity increases down Group 1 because as you move down How are. Thats why metals form positive ions and halogens form negative ions. It is important that you do all three they work together to make your revision. What this means is that Halogens GAIN electrons (theyre reduced) from the metals which LOSE electrons (theyre oxidised). Reactivity, unlike other periodic trends, does not increase or decrease going across a period or up and down a group. (NaOH) which is common chemical in laboratories and. Reactivity with water increases when going down the group. I know why group 1 reactivity increases as you go down the group but I am confused on group 7 and why it is different? Halogens are the opposite to group 1 and group 2 because 1&2 are metals and the halogens are non metals. Group 1 elements (alkali metals) readily reacts with water to produce metal hydroxide and hydrogen gas. What happened to reactivity down the group 17 The chemical reactivity of group 17 elements decreases down the group. In case of a group, reactivity increases on going down since the tendency to lose electrons increases but for non-metals, reactivity decreases on going down. As you move down the group, the amount of electron shielding increases, meaning that the electron is less attracted to the nucleus. This again is caused by the increase in atomic radius (there are more orbitals). This is because group 7 elements react by gaining an electron. This causes the electrons to experience a greater force of attraction as you move along the period, making it easier for an atoms to gain more electrons to become ions.Can someone help me with my chemistry homework? my teacher also wants me to describe how the trend in reactivity of the halogens down the group differs from that of the alkali metals down the group. Electron affinity decreases as we proceed down a group. 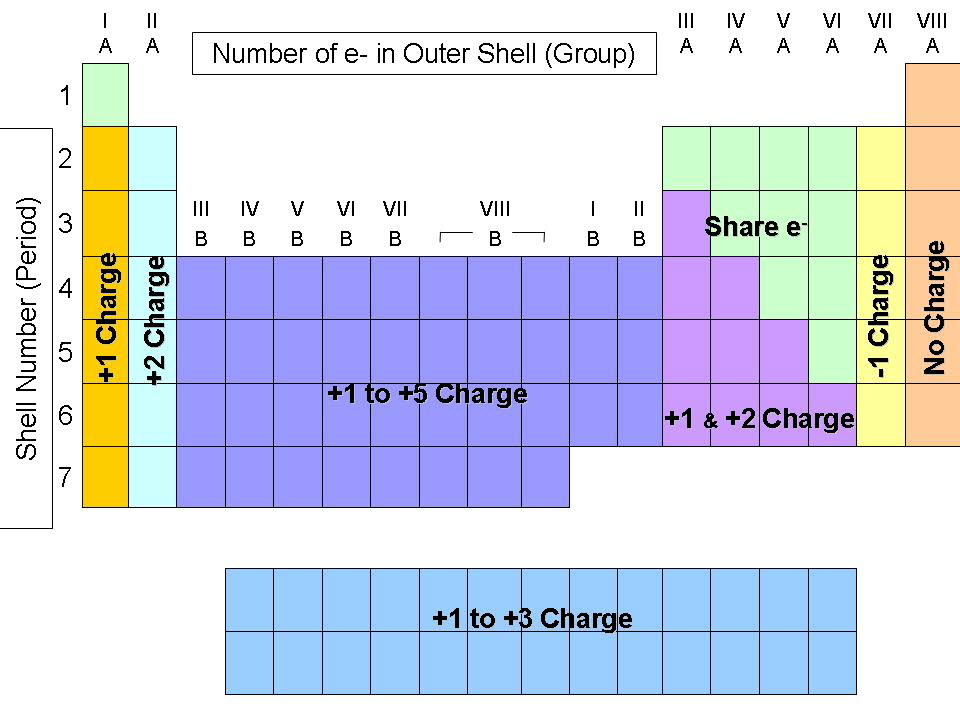
Reactivity of non-metals depends on the tendency to gain electrons. In case of metals the atomic size increases as we move down the table. If the atomic size increases, the valence electrons are easily removed thereby forming positive ion.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
